
Leveraging digital innovations to support the development of CRS de-risking products
Today, Cytokine Release Syndrome (CRS) requires regular clinical oversight through extended hospitalizations, creating barriers to care and limiting patient access to vital immunotherapies. Digital health technologies (DHTs) have matured to a point where we can—and should—rely on them to detect the onset of CRS, enabling monitoring and risk prediction beyond the clinic.
To support, DATAcc by DiMe has developed resources to support the development of trustworthy CRS de-risking products that can enable earlier detection and management, empowering oncologists to improve patient outcomes globally.
Immunotherapies have emerged as promising treatments for devastating hematological cancers like leukemias, lymphomas, and multiple myeloma. They offer targeted, better-tolerated options with promising results, such as improved survival rates and quality of life.
Cytokine Release Syndrome (CRS) can be a side effect of these immunotherapies and has become a significant risk for drug developers, healthcare providers, payors, regulators, and, most importantly, patients. It can threaten patients’ lives if the onset is not closely monitored and managed.
Who will benefit from these resources?

Developers

Providers

Patients
Building CRS De-risking Products: A Practical Guide for Developers
The guide provides you with actionable recommendations for developing CRS de-risking products, outlining the value proposition and key development milestones for bringing a safe and effective product to market.
Additional resources to support your success
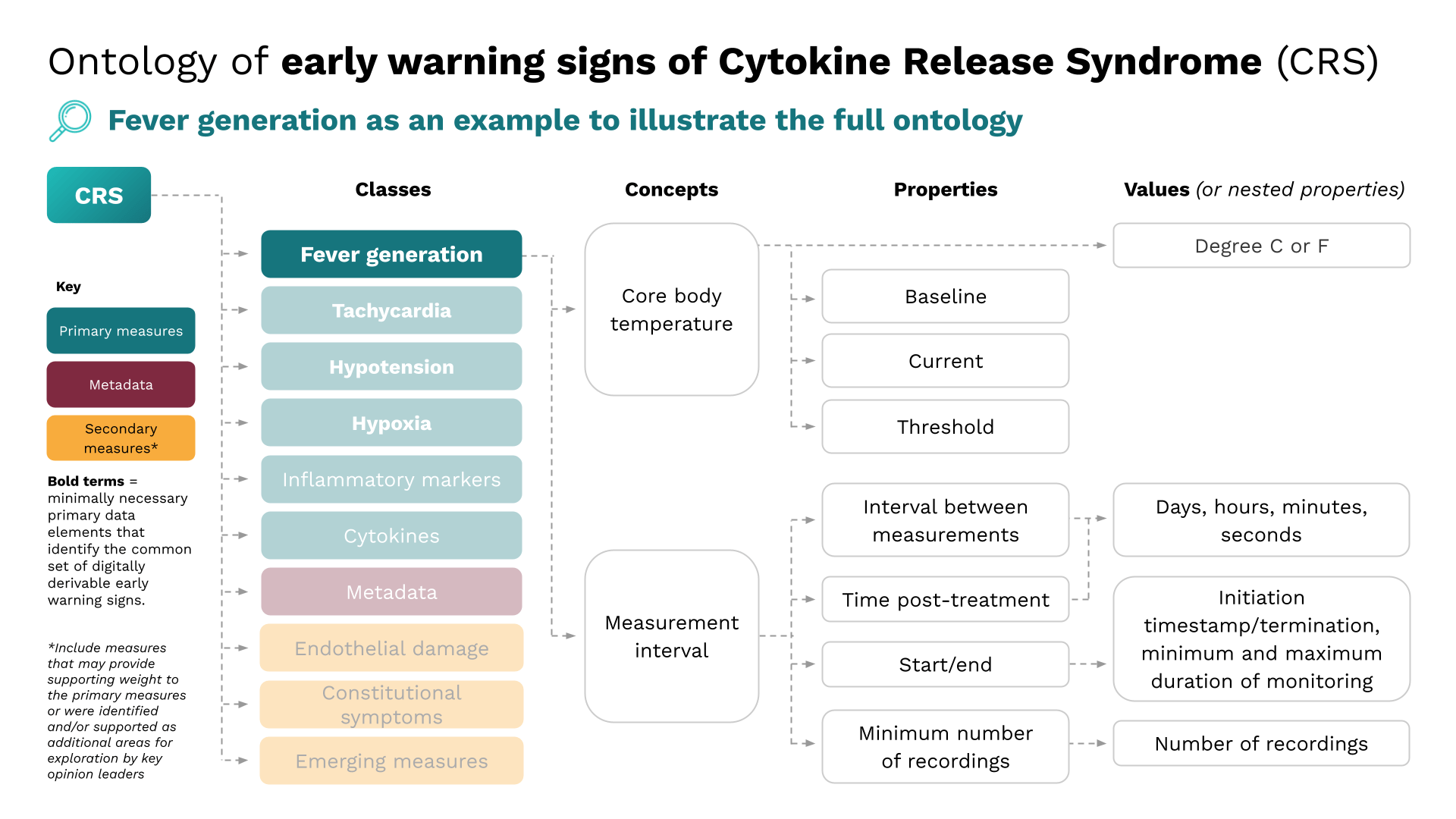
Digital clinical measures
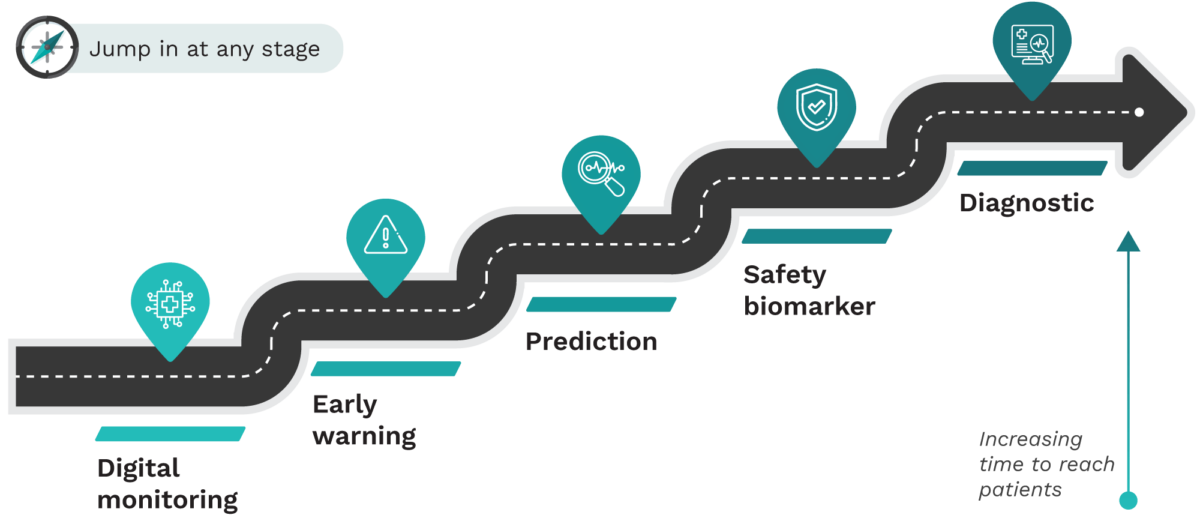
Innovation roadmap
Navigating CRS: The patient journey
The visual highlights Alexandra’s challenges and shows how a CRS de-risking product could have been used to improve her patient journey.
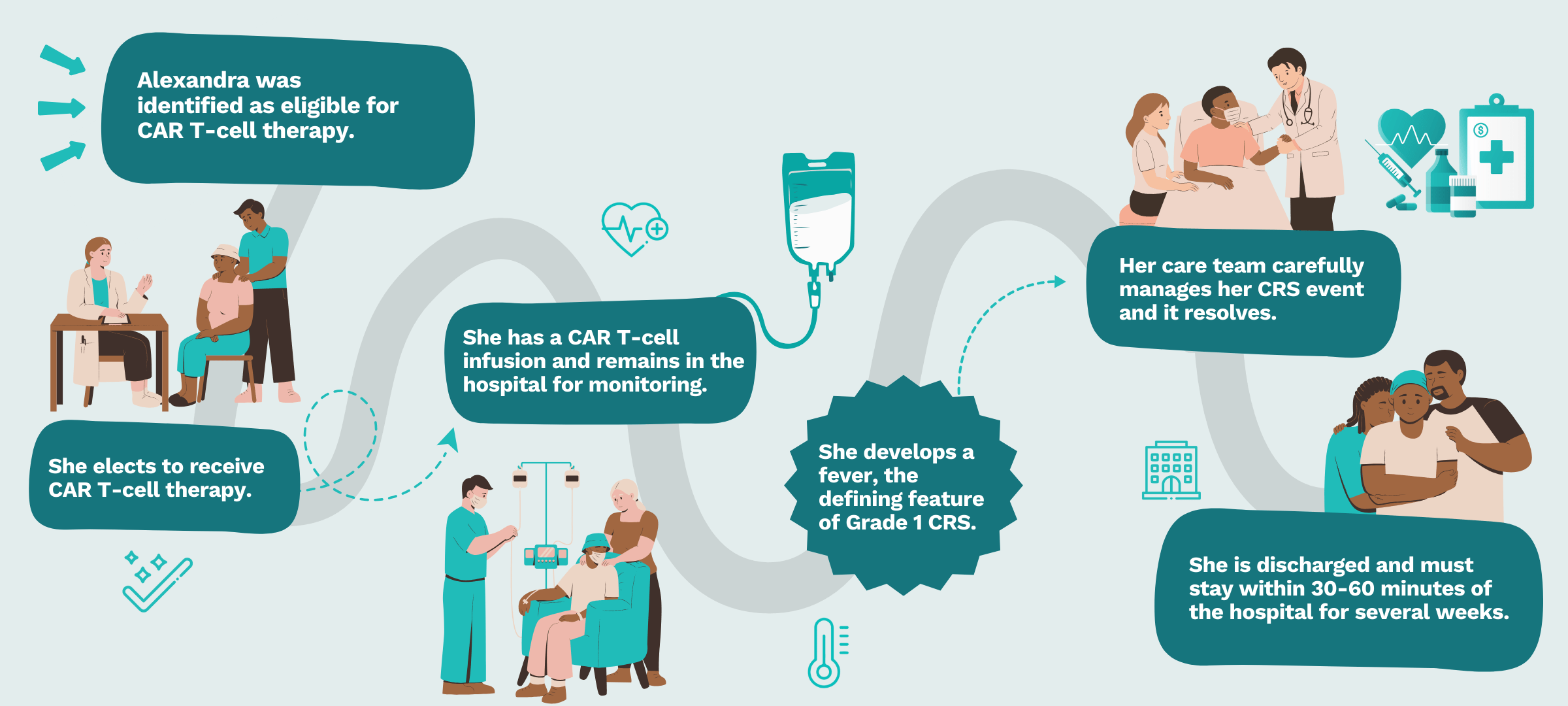
As immunotherapies, including those in development, become available sooner and indications expand, more patients like Alexandra will become eligible, challenging clinical staff to keep up with the growing demand for care.
Only 20% of eligible patients, like Alexandra, receive this therapy due to the complex logistics, toxicity concerns, and financial burden.
Her vitals are collected every four hours as standard of care.
Using DHTs could’ve enabled her CRS detection up to two hours earlier and supported monitoring in an outpatient setting.
Advanced CRS de-risking products could assess whether her CRS event would progress, allowing her team to manage the CRS event in the appropriate setting.
CRS de-risking products could allow Alexandra to safely distance herself from the clinic, potentially even returning home several hours away.
Discover how others are using these resources

In this clip, Nazila Shafagati of John’s Hopkins Medical Oncology emphasizes the importance of remote monitoring as a critical safety net for patients with lower health literacy or unreliable caregiving, helping to catch illness earlier and improve outcomes.

Cindy Varga, Atrium Health, highlights how a risk prediction model helps determine inpatient vs. outpatient care and identify patients at risk for CRS complications, emphasizing the importance of these tools.

Erik Koenig from Takeda highlights the need for standardized data transfer specifications and harmonization as a foundation for developing products applicable outside the hospital, enabling future modeling and predictive capabilities.

ActiGraph is pioneering the digital transformation of clinical research. We empower researchers to unleash the potential of big data and AI with a device-agnostic, decentralized trial platform backed by proven operational, scientific, and regulatory expertise.

“The right data to develop or validate a useful product depends on precisely understanding how it will be used, how it fits in existing clinical workflows, and its benefits to care providers and patients.
The DiMe CRS initiative brought together a diverse group of stakeholders to understand these perspectives better, providing invaluable insights about the types of data we need to gather to tackle immunotherapy patient monitoring. We will work closely with our partners and employ digital products to transform clinical research, gathering the right data to empower care providers and patients alike in this space.”
– Michael Pettinati | Senior Data Scientist, ActiGraph

“Early detection of Cytokine Release Syndrome (CRS) using digital health products and predictive algorithms will enable us to maximize the potential of cell therapies and T-cell engagers.
The collaboration with DATAcc by DiMe has been instrumental in supporting the industry towards this objective. I am optimistic about a future where more patients will have access to and benefit from these transformative therapies.”
– Premal Kamdar | Director, Digital Health Product Management, Bristol Myers Squibb

“Removing barriers to the adoption of novel and potentially game-changing therapies is at the core of what we do at Canopy. We’re proud to partner with the DATAcc by DiMe and other leaders to turn that vision into reality.”
– Lavi Kwiatkowsky | Founder & CEO, Canopy

“The Digital Clinical Measures for CRS will be a key reference for tailoring ADI’s RPM solution to capture the right data. They will help align our technology with clinical needs, delivering relevant insights to enhance patient care. Participation in the DATAcc by DiMe project has enriched our understanding of CRS management, thanks to the multidisciplinary expertise of the working group.”
-Milos Todorovic | Director of Biomedical and Clinical Engineering, Analog Devices, Inc.

“Leveraging DiMe’s ontology for data transfer specifications and standardizing data collection ensures uniform collection of all variables and enhances the reliability and comprehensiveness of our study observations.
This advancement paves the way for future research, ensuring Takeda remains at the forefront of innovative healthcare solutions.”
-Melinda Chen | Senior Scientist, Digital Biomarker Lead, Oncology Precision Translational Medicine, Takeda
-Erik Koenig | Head, PTM Strategic Innovation, Takeda

Takeda is a patient-focused, values-based, R&D-driven global biopharmaceutical company committed to bringing people better health and a brighter future worldwide.
More about this project

Moving the bar(rier) forward: the benefits of de-risking cytokine release syndrome

Advancing CRS care: Blue Spark Technologies’ role in early detection and innovation with DATAcc

Breaking Barriers with Digital Health Technologies: Advancing CRS Risk Prediction

De-risking CRS: Transforming care with digital technology





































